 
So, the molecule’s net dipole moment is zero, and it is non-polar. Even though each of the four C-Cl bonds is distinctly polar, the resultant moment of any three of them is equal in magnitude but opposite in direction to the moment due to the fourth one. Is CCl4 a dipole?Carbon tetrachloride,CCl4, has a net dipole moment of zero. In fact, Wikipedia writes: “Because of this small difference in electronegativities, the C−H bond is generally regarded as being non-polar.” Is CC more polar than CH?The C−H bond is indeed slightly more polar than the C−C bond, but this slightly increased polaity can in no solely way account for the approximately 20% increase in bond energy. … And also the polarity also depends on bond length and since due to the greater size of Cl atom the bond length is less than F and hence the low polarity. Why is C CL more polar than CF?The C-F bond is more polar than C-Cl bond because F is more electronegative than Cl. Element Electronegativity (Pauling) Cl 3.2 N 3.0 Br 3.0 I 2.7 9 more rows … Carbon is More Electronegative Than You Think. Iodine Pentafluoride, IF5 Molecular Geometry & Polarity H2O Lewis structure. Is CL more electronegative than C?Check out boron: it’s less electronegative than hydrogen. separation of charge occurs then a bond dipole moment is developed. However, bond polarity alone does not make a molecule polar. As a result, the entire molecule is nonpolar. For a bond to be polar, the electronegativity difference between the two atoms should be greater than or equal to 0.5. 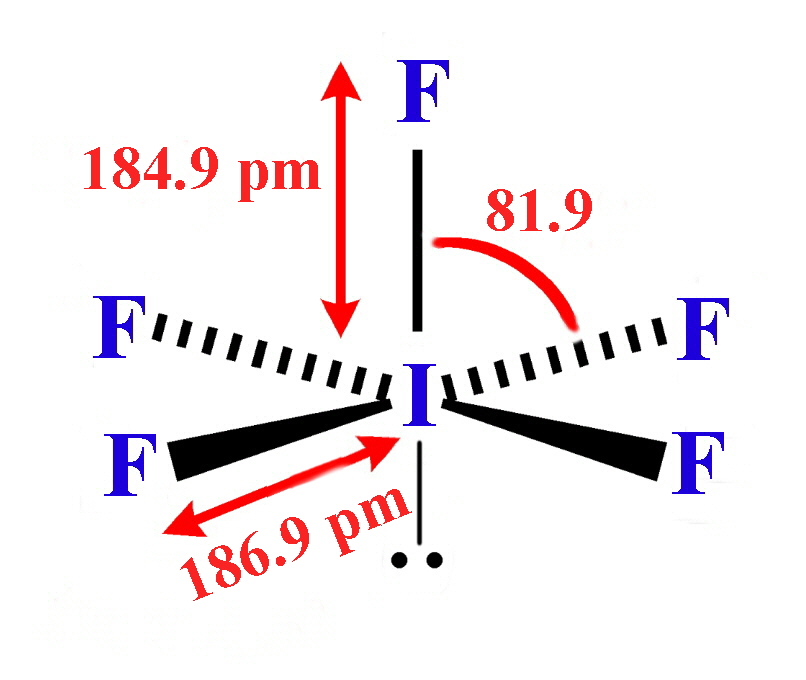
The C-H bond is also nonpolar because of nearly the same electronegativity. Ethylene (C2H4) is a linear-shaped molecule with a double bond between both carbon atoms (C=C). Burdick & Jackson solvents are arranged in order of increasing polarity index, a relative measure of the degree of interaction of the solvent with various polar test solutes. For heteronuclear molecules such as CO, there is a small dipole moment. Homonuclear diatomic molecules such as Br 2 and N 2 have no difference in electronegativity, so their dipole moment is zero. Is C2H4 polar or nonpolar molecule?Conclusion. For diatomic molecules, there is only one bond, so its bond dipole moment determines the molecular polarity. polar covalent bonds with partial negative charges on the I atoms. However, the molecular geometry of IF5 looks square pyramidal-shaped. The center Iodine atom of IF5 has one lone pair of electrons, resulting in square pyramidal IF5 electron geometry. This is the best answer based on feedback and ratings. In the IF5 molecular geometry, the I-F bonds have stayed in the five terminals and one lone pair of electrons on the Iodine atom of the square pyramidal molecule. polar covalent bonds with partial negative charges on the F atoms. nonpolar polar (b) IF5 polar nonpolar (c) SF6 nonpolar polar (d) SeBr4 polar nonpolar (e) SO2 polar nonpolar (f) O3 polar nonpolar. It is a polar molecule because it can have dipole moments due to uneven charges. Youll get a detailed solution from a subject matter expert that helps you learn core concepts. Is ammonia polar or nonpolar?Ammonia is a polar molecule: The electrostatic potential clearly shows that the nitrogen is partially negative whereas the hydrogens are partially positive. IF5 is considered a type of polar molecule. Electronegativity is the measure of how strongly an atom will attract electrons to itself. 
Is carbon disulfide polar or nonpolar?Carbon disulfide is not a polar molecule. CS2: the molecule has a linear geometry and the electronegativity difference between C and S is 0, so this one is not polar either. 
In addition, the attractive interaction between dipoles falls off much more rapidly with increasing distance than do the ion–ion interactions.Is BrCl3 polar or nonpolar?BrCl3: the molecule has a T-shaped molecular geometry which could make it polar, but the Br-Cl electronegativity difference is 0.2 which makes it a covalent bond. \): Both Attractive and Repulsive Dipole–Dipole Interactions Occur in a Liquid Sample with Many Moleculesīecause each end of a dipole possesses only a fraction of the charge of an electron, dipole–dipole interactions are substantially weaker than the interactions between two ions, each of which has a charge of at least ☑, or between a dipole and an ion, in which one of the species has at least a full positive or negative charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
